HYPE® Acetabular Cup
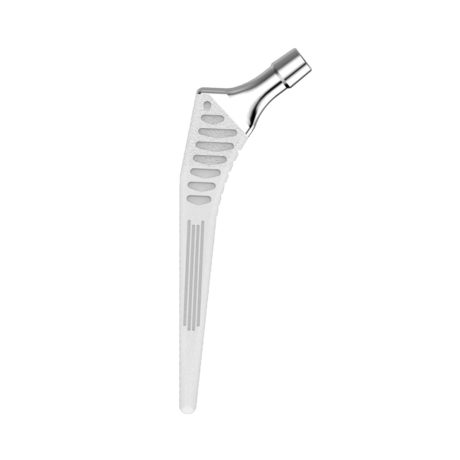
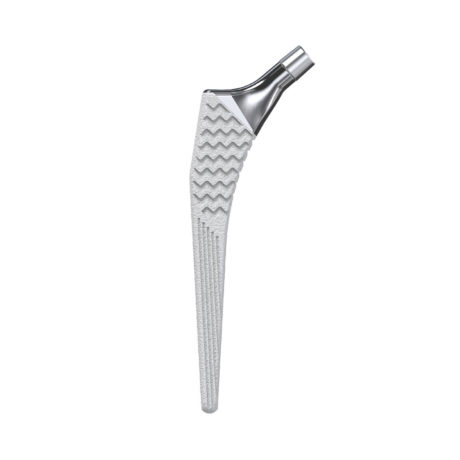
The Hype® acetabular cup is an implant dedicated to primary total hip arthroplasty with a cementless fixation. The cup is designed to be combined with either a Biolox® delta ceramic liner or an UHMWPE liner.
Fixing screws (optional) of Ø 6.5 mm are available to secure the cup if required due to the bone quality. These screws are available in lengths ranging from 20 to 45 mm (in 5-mm increments).
This implant is part of the Hype® femoral stem range.
Characteristics and materials
Characteristics:
- The Hype® cup is available in diameters of 45 mm to 63 mm.
- The HIC ceramic liner is intended for Ø 28, 32 and 36 mm femoral heads.
- The HIPER liner has an overhang and is intended for Ø 22.2 and 28 mm femoral heads.
- Fixing screws (optional) have a diameter of 6.5 mm and are available in a length of 20 to 45 mm (in 5 mm increments).
Materials:
- The Hype® cup is made from titanium alloy + dual coating made of titanium spray and HA.
- The screws are made from titanium alloy.
- The HIC insert is made from Biolox® delta ceramic.
- The HIPER insert is made from UHMWPE.
Instruments
- One instrumentation set for the two versions of Hype® liner
- Additional sets available:
- for preparation and insertion of the locking screws.
- for anterior approach and minimally invasive surgeries.
Indications
The Hype® cup is indicated in the following cases:
- Primary or secondary arthrosis,
- Inflammatory degenerative joint diseases,
- Fractures of the proximal femur,
- Avascular necrosis
- These indications are appropriate according to the following classifications: step 1, 2, 3 of SOFCOT or type 1, 2A, 2B, 2C of Paprosky classification.
- The use of ceramics appears to be particularly suited to young active patients (reduction in the degree of wear compared with conventional friction torque).
- Several inflammatory degenerative joint diseases may lead to a pathological hip. Some of them are listed hereunder. However, this list is informative:
- Rheumatoid arthritis,
- Spondylitis.
Contraindications for the Hype® cup are as follows:
- Localized or systemic acute or chronic infection,
- Skeletal immaturity,
- Pregnant or nursing women,
- Known allergy to one of the prosthesis components (see figure 6),
- Severe muscular, neurological, or vascular deficiency affecting the concerned limb,
- Bone degradation or bone loss, or poor bone quality likely to affect the stability of the implant,
- Local bone tumors,
- Obesity.